DDT can be prepared by reacting chlorobenzene in the presence of conc.\[{H_2}S{O_4}\] with ______.
Answer
255k+ views
Hint: The full form of DDT is dichlorodiphenyltrichloroethane. DDT is a colourless, odourless, tasteless, crystalline chemical compound. The compound to which it reacts is an aldehyde.
Complete Step by Step Solution:
DDT or dichlorodiphenyltrichloroethane is used as an insecticide in agriculture. The chemical formula of DDT is \[{C_{14}}{H_9}C{l_5}\] . The compound is present as a colourless and tasteless crystalline solid in its standard condition for temperature and pressure.
DDT is prepared by reacting chloral and chlorobenzene in presence of concentrated sulphuric acid in a ratio of 1:2.
Chloral is an organic compound having a molecular formula of \[C{l_3}CCHO\]. The other name of chloral is trichloroacetaldehyde or trichloroethanal. It is a colourless oily aldehyde compound which shows solubility in most solvents.
The reaction between chlorobenzene and chloral is shown below.
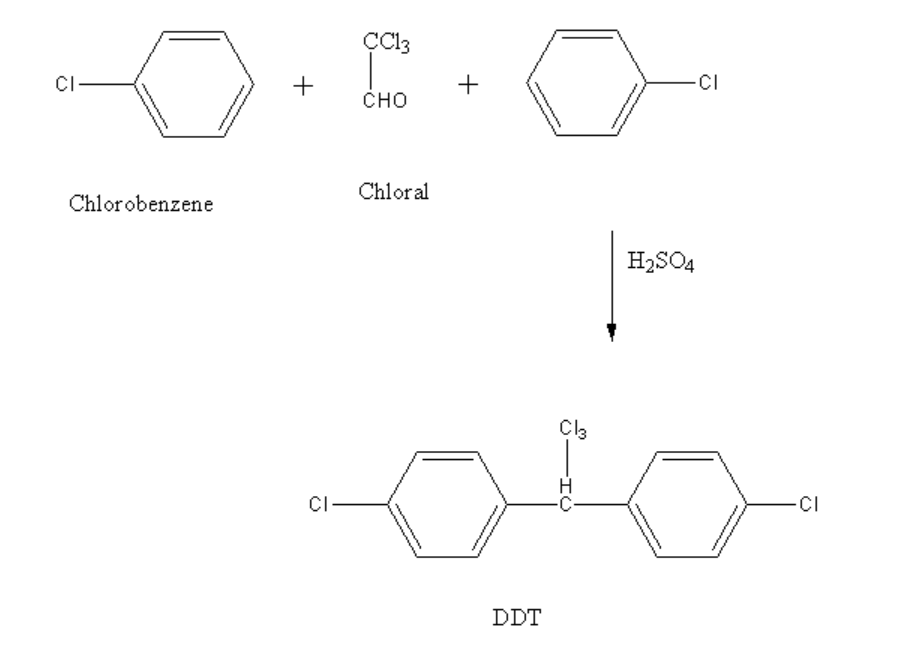
Image: Preparation of DDT
In the above reaction, two moles of chlorobenzene reacts with one mole of chloral in presence of concentrated sulphuric acid to form dichlorodiphenyltrichloroethane.
Therefore, DDT can be prepared by reacting chlorobenzene in the presence of conc.\[{H_2}S{O_4}\] with chloral.
Additional information: DDT is now banned in different countries as it is a very toxic compound and due to its high usage it leads to death. Chloral on dissolving in water forms chloral hydrate.
Note: Sometimes it may be confusing while drawing the structure of DDT as it is a germinal compound where two benzyl chloride group is attached to one carbon and only one chlorine group is on the other carbon atom.
Complete Step by Step Solution:
DDT or dichlorodiphenyltrichloroethane is used as an insecticide in agriculture. The chemical formula of DDT is \[{C_{14}}{H_9}C{l_5}\] . The compound is present as a colourless and tasteless crystalline solid in its standard condition for temperature and pressure.
DDT is prepared by reacting chloral and chlorobenzene in presence of concentrated sulphuric acid in a ratio of 1:2.
Chloral is an organic compound having a molecular formula of \[C{l_3}CCHO\]. The other name of chloral is trichloroacetaldehyde or trichloroethanal. It is a colourless oily aldehyde compound which shows solubility in most solvents.
The reaction between chlorobenzene and chloral is shown below.

Image: Preparation of DDT
In the above reaction, two moles of chlorobenzene reacts with one mole of chloral in presence of concentrated sulphuric acid to form dichlorodiphenyltrichloroethane.
Therefore, DDT can be prepared by reacting chlorobenzene in the presence of conc.\[{H_2}S{O_4}\] with chloral.
Additional information: DDT is now banned in different countries as it is a very toxic compound and due to its high usage it leads to death. Chloral on dissolving in water forms chloral hydrate.
Note: Sometimes it may be confusing while drawing the structure of DDT as it is a germinal compound where two benzyl chloride group is attached to one carbon and only one chlorine group is on the other carbon atom.
Recently Updated Pages
JEE Main 2026 Exam Date, Session 2 Results OUT, Cutoff Released, City Slip & Admit Card

JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

Trending doubts
JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding Atomic Structure for Beginners

Understanding Electromagnetic Waves and Their Importance

Inductive Effect and Its Role in Acidic Strength

Degree of Dissociation: Meaning, Formula, Calculation & Uses

Other Pages
JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

Understanding the Electric Field Due to Infinite Linear Charge and Cylinders

Understanding Average and RMS Value in Electrical Circuits

DDT can be prepared by reacting chlorobenzene in the class 12 chemistry JEE_Main

Is PPh3 a strong ligand class 12 chemistry JEE_Main

Common Ion Effect: Concept, Applications, and Problem-Solving




