



What is Haemoglobin Formula and Composition?
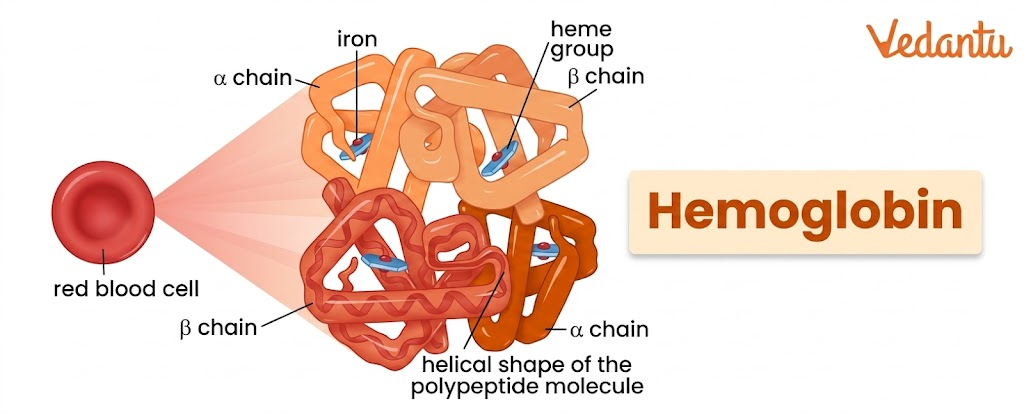
Haemoglobin is an iron-containing respiratory pigment and globular protein present mainly in red blood cells. Its main job is to transport oxygen from the lungs to body tissues and carry a part of carbon dioxide back to the lungs. It is a tetrameric protein composed of four globin chains, and each chain is attached to an iron-containing heme group. Because of this structure, one haemoglobin molecule can bind up to four oxygen molecules.
The term haemoglobin is commonly abbreviated as Hb.
It is a globular protein with a highly specialised structure that allows it to bind oxygen reversibly.
This reversible binding is important because haemoglobin must pick up oxygen in the lungs and release it in tissues where oxygen is needed.
Haemoglobin does much more than simply carry oxygen.
It also carries a portion of carbon dioxide from tissues back to the lungs and helps in maintaining efficient gas exchange.
In addition, it can bind nitric oxide and participate in the physiological regulation of blood flow in certain contexts.
Where is Haemoglobin Found?
Haemoglobin is found mainly inside red blood cells or erythrocytes. These cells are specifically designed to transport respiratory gases, and haemoglobin accounts for a very large proportion of their dry weight.
In fact, about 90 to 95 per cent of the dry weight of red blood cells is haemoglobin, underscoring its importance for RBC function.
Haemoglobin develops in the cells of the bone marrow while red blood cells are being formed.
During maturation, precursor cells synthesise haemoglobin in large amounts.
Even after the nucleus is lost in mammalian RBC development, some haemoglobin synthesis can continue in reticulocytes for a short period before they become fully mature erythrocytes.
Haemoglobin Structure
Haemoglobin is a tetrameric protein, which means it is made up of four polypeptide subunits. In the major adult form of haemoglobin, these four chains are arranged as:
two alpha chains
two beta chains
So, the main adult structure of haemoglobin is written as α2β2.
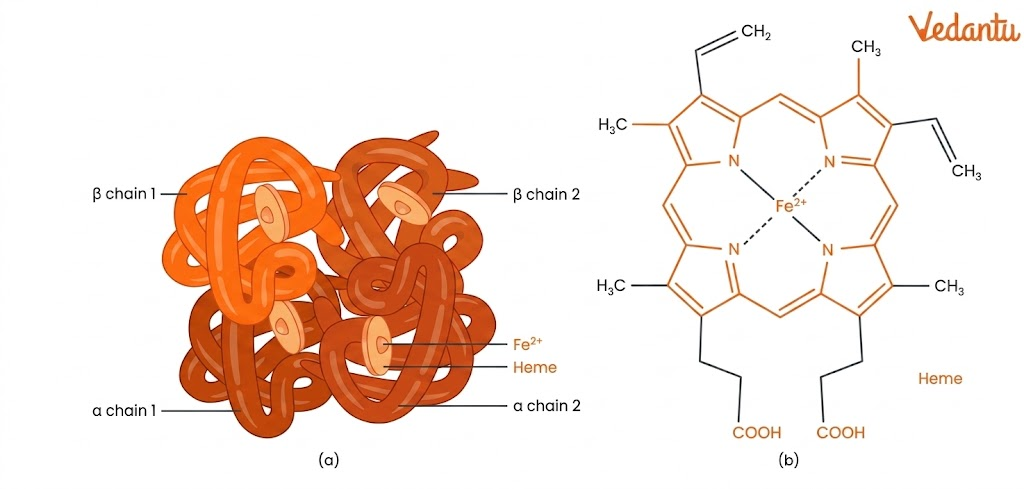
Each polypeptide chain is associated with one heme group. Since there are four globin chains, one haemoglobin molecule contains four heme groups. Each heme group contains a central iron atom that can bind one oxygen molecule. Therefore, one haemoglobin molecule can bind a maximum of four oxygen molecules.
Globin Part
The protein part of haemoglobin is called globin. In adult haemoglobin:
Each alpha chain contains 141 amino acids
Each beta chain contains 146 amino acids
These chains fold into a compact globular form. Most of their amino acids form alpha helices connected by short non-helical segments. This gives haemoglobin its stable three-dimensional structure.
Heme Part
The non-protein part of haemoglobin is the heme group, which is an iron-containing prosthetic group. The iron atom lies at the centre of a porphyrin ring made of nitrogen-containing structures. This iron is the site where oxygen binds.
A very important point about NEET is that oxygen does not bind directly to the protein chain. It binds to the iron atom in the heme group.
Quaternary Structure
Haemoglobin has a well-defined quaternary structure because the four subunits associate with one another through:
hydrophobic interactions
ionic interactions or salt bridges
There is a particularly strong interaction between the alpha and beta subunits. On mild treatment, haemoglobin may partially dissociate, but alpha-beta dimers tend to remain intact.
R State and T State
Another important part of the haemoglobin structure is that it can exist in two functional conformations:
T state or tense state
R state or relaxed state
The T state has lower affinity for oxygen and is associated mainly with deoxyhaemoglobin. The R state has a higher affinity for oxygen and is associated with oxygen binding. The switch between these two states underlies cooperative oxygen binding.
Function of Haemoglobin
Its main role is to transport oxygen from the lungs to the tissues. However, this is not its only function.
1. Transport of Oxygen
The primary function of haemoglobin is oxygen transport. In the lungs, where the partial pressure of oxygen is high, haemoglobin binds oxygen and forms oxyhaemoglobin. This oxygen-rich blood then circulates to the body tissues.
In tissues, where oxygen concentration is lower and carbon dioxide concentration is higher, haemoglobin releases oxygen. This ensures that cells receive oxygen for respiration and energy production.
Each heme group can bind one oxygen molecule, so one haemoglobin molecule can carry up to four oxygen molecules.
2. Transport of Carbon Dioxide
Haemoglobin also helps transport carbon dioxide. Around 20 to 25 per cent of carbon dioxide is carried in the blood bound to haemoglobin as carbaminohaemoglobin. In tissues, where the carbon dioxide level is high, binding is favoured. In the lungs, carbon dioxide dissociates and is exhaled.
The remaining carbon dioxide is mostly transported in the blood as bicarbonate ions.
3. Nitric Oxide Transport
Haemoglobin can also transport nitric oxide bound to globin protein chains. This has regulatory significance in the body.
4. Contribution to Efficient Gas Exchange
The design of haemoglobin allows it to load oxygen quickly in the lungs and unload it efficiently in tissues. This makes it far more effective than simple dissolved oxygen transport.
How Oxygen Binds to Haemoglobin?
Oxygen binds reversibly to the iron atom present in each heme group. This binding is called oxygenation.
Cooperative Binding
Haemoglobin shows cooperative binding. This means that when one oxygen molecule binds to haemoglobin, the affinity of the remaining subunits for oxygen increases. As a result:
The first oxygen binds with relative difficulty
The second and third bind more easily
By the time the fourth oxygen binds, haemoglobin is in a high-affinity state
This is why the oxygen dissociation curve of haemoglobin is sigmoid or S-shaped.
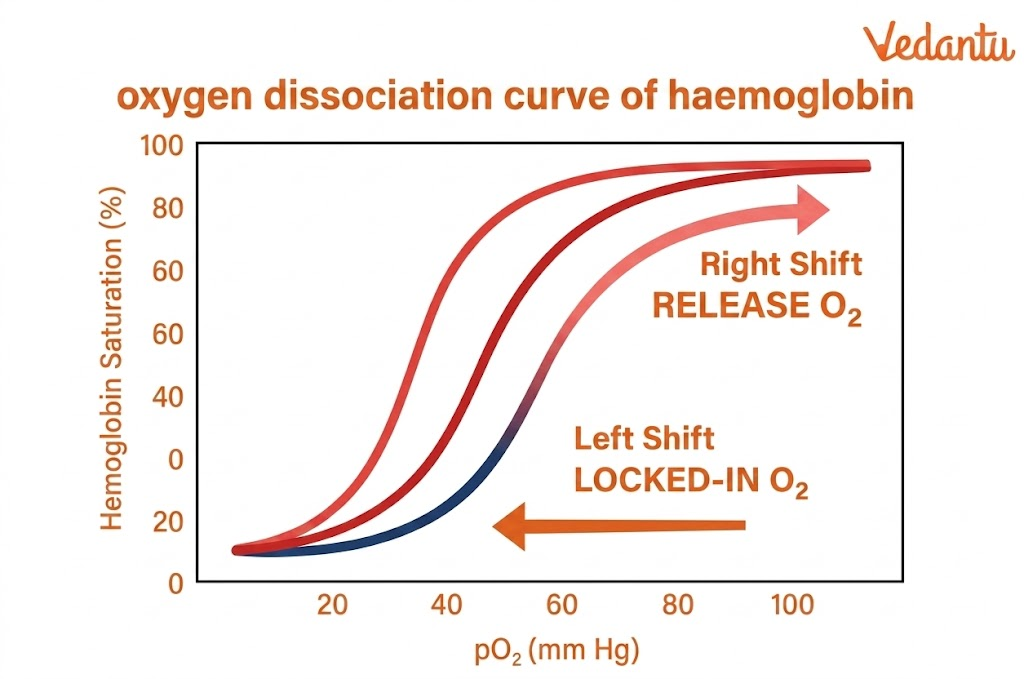
Factors Affecting Oxygen Binding
The affinity of oxygen to haemoglobin is affected by:
partial pressure of oxygen
pH
carbon dioxide concentration
2,3-BPG
In the lungs:
pO2 is high
pCO2 is low
pH is relatively higher
These conditions favour oxygen binding.
In tissues:
pO2 is low
pCO2 is high
pH is lower
These conditions favour oxygen release.
This explains why haemoglobin loads oxygen in the lungs and unloads it in the tissues.
How Haemoglobin is Formed?
Haemoglobin synthesis begins in developing red blood cell precursors in the bone marrow. It starts in proerythroblasts and continues into the reticulocyte stage.
A simplified sequence of haemoglobin formation is:
Step 1: Formation of Pyrrole
Succinyl-CoA from the Krebs cycle combines with glycine to form a pyrrole molecule.
Step 2: Formation of Protoporphyrin IX
Four pyrrole molecules join to form protoporphyrin IX.
Step 3: Formation of Heme
Iron combines with protoporphyrin IX to form the heme molecule.
Step 4: Formation of Globin Chains
Ribosomes synthesise long polypeptide chains called globin chains in the cytoplasm.
Step 5: Formation of Haemoglobin Subunits
Each heme molecule associates with one globin chain to form a haemoglobin subunit.
Step 6: Formation of Complete Haemoglobin
Four such subunits combine loosely to form one complete haemoglobin molecule.
This is why iron is an essential dietary mineral. Without enough iron, proper haemoglobin synthesis cannot occur.
Types of Haemoglobin
The body contains different types of haemoglobin, depending on age and globin chain composition.
Haemoglobin A
This is the most common haemoglobin in adults. Its composition is:
α2β2
It accounts for about 95-98 per cent of haemoglobin in normal adults.
Haemoglobin A2
This form is present in smaller amounts in adults. Its composition is:
α2δ2
It usually forms about 2 to 3 per cent of adult haemoglobin.
Haemoglobin F
This is fetal haemoglobin, found mainly in the fetus and newborns. Its composition is:
α2γ2
Fetal haemoglobin has a higher affinity for oxygen than adult haemoglobin, which helps the fetus obtain oxygen from maternal blood.
Other Variants
Other forms, such as haemoglobin H, may be seen in pathological conditions. Variations in globin chain structure can lead to haemoglobinopathies.
Haemoglobin Normal Range
The haemoglobin normal range varies with age and sex.
A broad healthy adult range is usually 12 to 20 g/dL, but normal reference values differ by sex, age, and developmental stage.
General Reference Values
Newborn: 17 to 22 g/dL
Children: 11 to 13 g/dL
Adult male: about 13.5 to 17.5 g/dL
Adult female: about 12 to 15.5 g/dL
Older males: around 12.4 to 14.9 g/dL
Older females: around 11.7 to 13.8 g/dL
These values help doctors assess whether a person may have anaemia or abnormally high haemoglobin.
Normal Haemoglobin Level in Male and Female
The normal haemoglobin level is generally higher in males than in females.
1. The usual haemoglobin range in male adults is around:
13.5 to 17.5 g/dL
Some references may show it slightly differently, but this range is commonly accepted in physiology-based explanations.
2. The usual haemoglobin level in female adults is around:
12 to 15.5 g/dL
What Would Be the Consequences of a Deficiency of Haemoglobin in Our Body?
Haemoglobin deficiency reduces the oxygen-carrying capacity of blood. If blood cannot carry enough oxygen, tissues do not receive the oxygen they need for proper cellular respiration. As a result, the body begins to show signs of poor oxygen supply.
Major consequences include:
tiredness and weakness due to poor energy production
reduced physical stamina
pale appearance because of low RBC haemoglobin content
shortness of breath due to inadequate oxygen delivery
dizziness or light-headedness
poor concentration and reduced alertness
A faster heartbeat occurs in severe cases because the body tries to compensate.
In prolonged deficiency, anaemia and organ stress may occur
So, the consequences of haemoglobin deficiency are directly related to impaired oxygen transport.
Low Haemoglobin Symptoms
Low haemoglobin is commonly associated with anaemia.
Common symptoms include:
weakness
fatigue
dizziness
shortness of breath
pale or yellowish skin
chest discomfort in severe cases
headache
poor concentration
Reduced exercise tolerance
The exact symptoms depend on how low the haemoglobin level is and how quickly it has fallen.
Diseases Related to Haemoglobin
Abnormal haemoglobin can cause serious disorders called haemoglobinopathies.
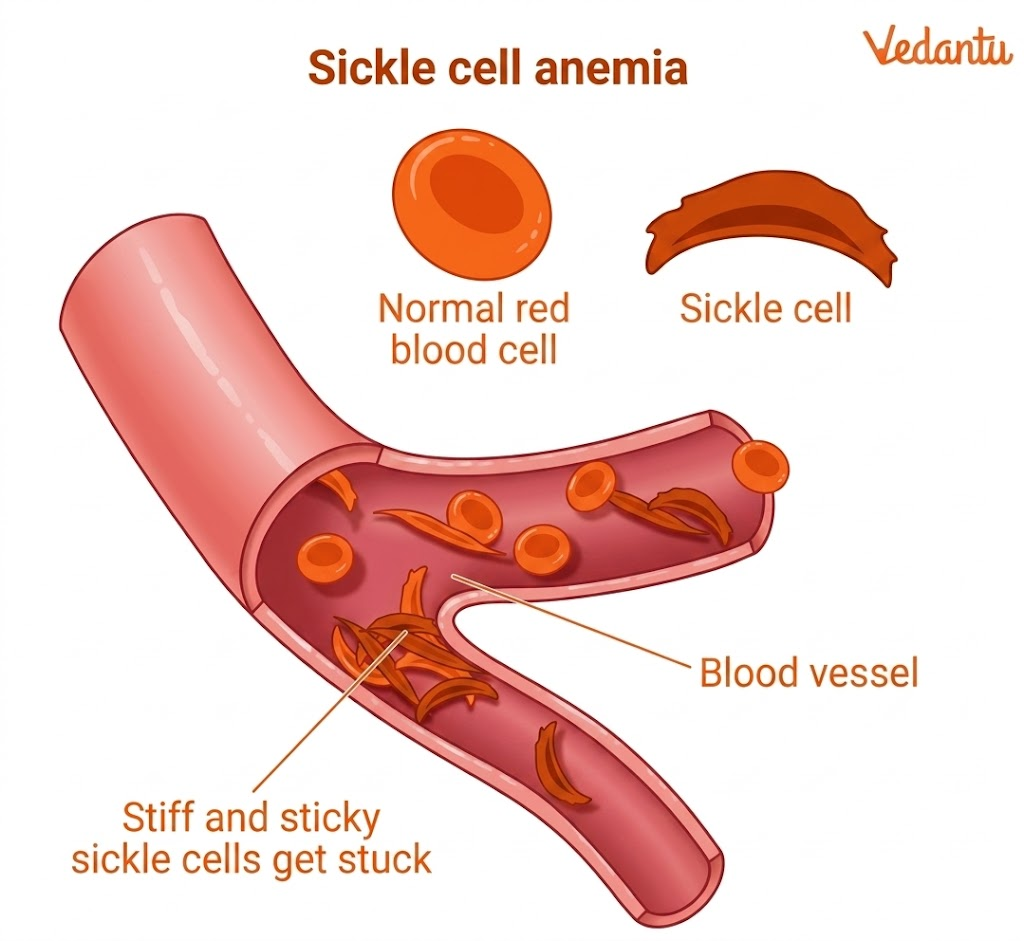
Sickle Cell Anaemia
A mutation in the beta globin gene causes sickle cell anaemia. A single nucleotide change leads to the replacement of glutamic acid by valine at the sixth position of the beta chain. This changes the properties of haemoglobin and causes RBCs to become sickle-shaped under certain conditions.
These abnormal RBCs can block blood vessels and carry oxygen less efficiently.
Thalassaemia
Thalassaemia is caused by reduced synthesis of globin chains. It may involve:
alpha-thalassaemia
beta-thalassaemia
The severity depends on how many genes are defective or missing.
Abnormal Haemoglobin Variants
Mutant or abnormal haemoglobin variants may also occur, such as:
Haemoglobin S
Haemoglobin C
Haemoglobin E
These can lead to varying levels of haemolytic anaemia or related blood disorders.
Glycosylated Haemoglobin
HbA1c is glycosylated haemoglobin and is used clinically to monitor average blood glucose levels in diabetic patients.
Haemoglobin-Rich Foods
Foods themselves do not contain haemoglobin in the way blood does, but many foods help the body make more haemoglobin because they provide iron, folate, vitamin B12, protein, and other nutrients.
Common haemoglobin-rich foods or foods that support haemoglobin production include:
green leafy vegetables such as spinach
beetroot
pomegranate
dates and raisins
jaggery
pulses and legumes
beans
nuts and seeds
meat, liver, fish, and eggs
iron-fortified cereals
vitamin C-rich fruits such as oranges and amla, which help improve iron absorption
How to Increase Haemoglobin?
Eat iron-rich foods regularly.
Include vitamin C-rich foods to improve iron absorption
Ensure enough folate and vitamin B12 in the diet
Take medical treatment if iron deficiency anaemia is diagnosed
Treat underlying causes such as chronic disease, blood loss, or nutritional deficiency
Maintain a balanced protein-rich diet because globin is a protein
If haemoglobin is severely low, medical evaluation is necessary. Dietary correction alone may not always be enough.
Significance of Haemoglobin in Blood
Haemoglobin has enormous physiological significance because it allows blood to transport oxygen efficiently. Without haemoglobin, the amount of oxygen dissolved directly in plasma would be far too little to meet body needs.
Its significance includes:
efficient oxygen delivery to tissues
partial transport of carbon dioxide
contribution to acid-base balance indirectly through gas transport
support of normal tissue respiration
diagnostic value in blood tests
What Experts Have to Conclude?
Haemoglobin is one of the most important proteins in human physiology. It is the chief respiratory pigment of red blood cells and plays a central role in oxygen transport, carbon dioxide transport, and efficient gas exchange. To master this topic, focus on the following high-yield areas:
tetrameric structure of haemoglobin
role of iron and heme
cooperative binding of oxygen
R state and T state
normal haemoglobin ranges in males and females
Haemoglobin deficiency and its consequences
major haemoglobin disorders such as sickle cell anaemia and thalassaemia
This topic is highly relevant not only to NEET Biology but also to understanding blood physiology and respiratory transport in a deeper, more comprehensive way.
FAQs on Haemoglobin: Structure, Function, Normal Range, Deficiency and Foods for NEET
1. What happens when haemoglobin is low?
When haemoglobin is low, the blood carries less oxygen to the body. This can make a person feel tired, weak, dizzy, and short of breath. The skin may look pale, and hands and feet may feel cold. If the low haemoglobin level is left untreated, it can put stress on the heart and other organs.
2. How can I increase my haemoglobin?
To increase haemoglobin, focus on foods and habits that support blood formation.
Eat iron-rich foods like spinach, lentils, beans, dates, raisins, eggs, fish, and meat.
Add vitamin C-rich foods like oranges, lemons, amla, and tomatoes because vitamin C helps the body absorb iron better.
Also, make sure you get enough folate, vitamin B12, and protein.
Avoid tea or coffee with meals because they can reduce iron absorption.
If haemoglobin is very low, a doctor may recommend supplements or other treatments.
3. What are normal haemoglobin levels?
The normal haemoglobin level varies between sex and age. In adult males, the haemoglobin is usually 4.5 to 17.5 g/dL. In adult females, the haemoglobin level in females is usually around 12.0 to 15.5 g/dL. These values are often called the haemoglobin normal range.
4. What are the 4 types of haemoglobin?
The four commonly discussed types of haemoglobin are:
HbA – the main adult haemoglobin
HbA2 – a minor adult type
HbF – fetal haemoglobin found mainly in babies
HbS – an abnormal type linked with sickle cell disease
These types differ in their globin chains and function.
5. What fruits increase haemoglobin?
Some fruits can help increase haemoglobin by providing iron, folate, or vitamin C. Good examples are:
Pomegranate
Dates
Raisins
Apples
Figs
Oranges
Lemons
Kiwi
Guava
Strawberries
Vitamin C-rich fruits are especially useful because they help the body absorb iron better.
6. What is a dangerously low haemoglobin level?
A dangerously low haemoglobin level is usually defined as below 7 g/dL. At this level, the body may not get enough oxygen, and urgent medical treatment may be needed. If haemoglobin falls even lower, it can become life-threatening.
7. What are the 5 symptoms of anaemia?
Five common symptoms of anaemia are:
Fatigue
Pale skin
Shortness of breath
Dizziness
Heart palpitations
These happen when low haemoglobin reduces oxygen.
8. Which organ is most affected by anemia?
The heart is one of the most affected organs in anaemia. This is because it has to work harder to pump oxygen-poor blood to the body. Over time, severe anaemia can put a lot of strain on the heart.
The heart is one of the most affected organs in anaemia. This is because it has to work harder to pump oxygen-poor blood to the body. Over time, severe anaemia can place a significant strain on the heart.
9. What vegetables are high in iron?
Vegetables high in iron include:
Spinach
Kale
Beet greens
Swiss chard
Broccoli
Peas
Beans
Lentils
Sweet potatoes
Brussels sprouts
These are useful for people trying to improve haemoglobin, especially when eaten with vitamin C-rich foods.
10. What is the rule of 3 for haemoglobin?
The rule of 3 for haemoglobin is a simple way to check CBC values. It means:
Hematocrit is about 3 times the haemoglobin value
Haemoglobin is about 3 times the RBC count, in a rough way
This rule is mainly used as a quick check for consistency in lab results, not as a final diagnosis.























